|
Chemical reactions largely involve atoms or groups of atoms and the interactions between their electrons. In general, electrons are easier to add or remove from an atom than a proton or neutron. The particles within an atom are bound together by powerful forces.Isotopes and ions of an atom with a constant number of protons are all variations of a single element. Varying the number of electrons results in ions. Varying the number of neutrons results in isotopes. The number of protons (also known as its atomic number) determines the element.Almost all of the mass of an atom is in its nucleus almost all of the volume of an atom is occupied by electrons.Electrons move around outside the nucleus.The nucleus carries a positive electrical charge. The nucleus of an atom contains protons and neutrons. transmitted light synonyms, transmitted light pronunciation, transmitted light translation, English dictionary definition of transmitted light.The mass of a proton is 1840 times greater than the mass of an electron. The mass of a proton is essentially the same as that of a neutron.Protons and neutrons are about the same size as each other and are much larger than electrons.In other words, neutrons do not have a charge and are not electrically attracted to either electrons or protons. Electrons and protons are electrically attracted to each other. 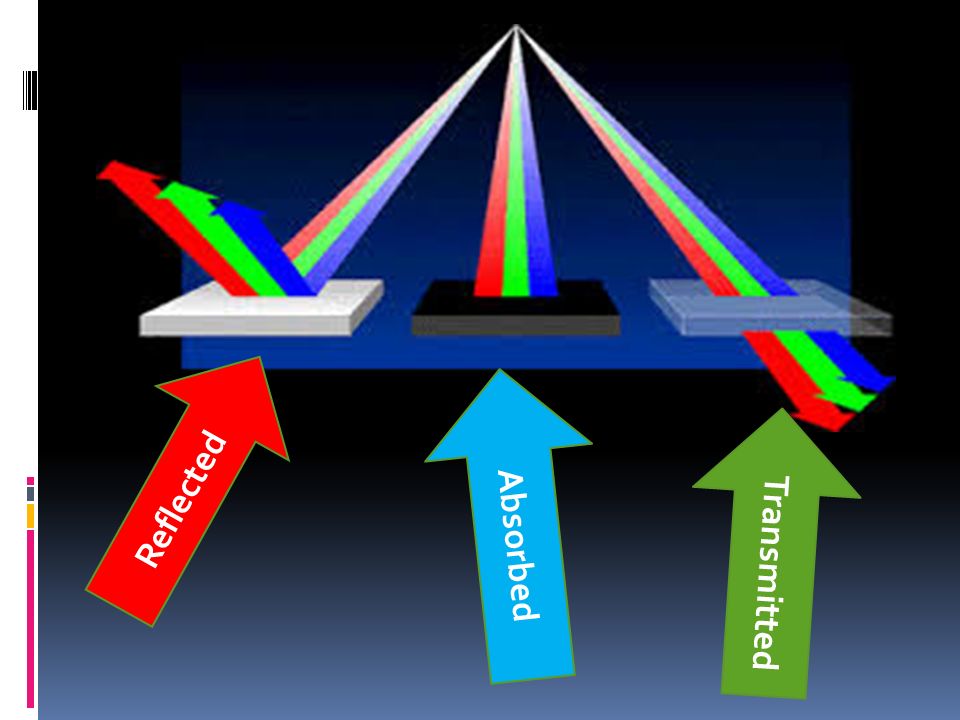
Absorption The absorptance Being ratios of radiant power values, reflectance. The charge of a proton and an electron are equal in magnitude, yet opposite in sign. Transmission is the passage of electromagnetic radiation through a medium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
